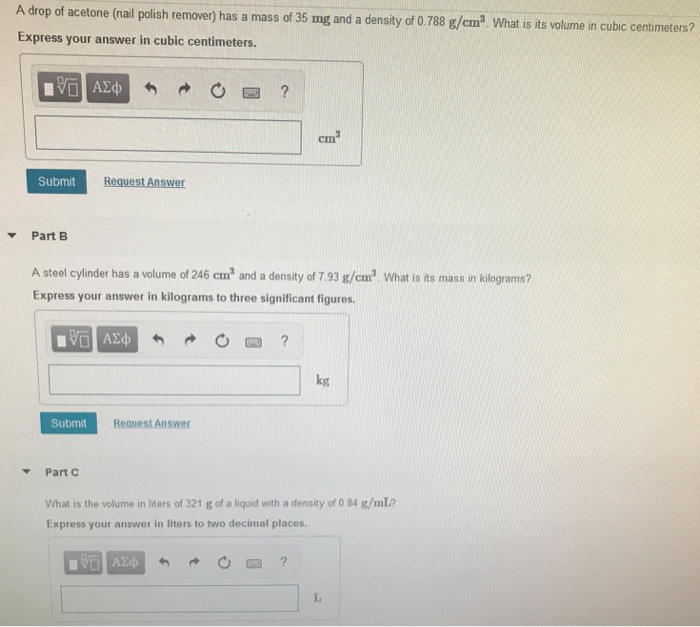
Acetone, the solvent in nail polish remover, has a density of 0791 g/mL What is the volume, in mil - YouTube

Thermophysical Properties of Liquid Acetone, Cellulose Acetate, and... | Download Scientific Diagram

SOLVED: The density of acetone is stated to be 791 kg/m^3, and 1 m^3 = 1*10^6 cm^3. what is the density of acetone in g/cm^3
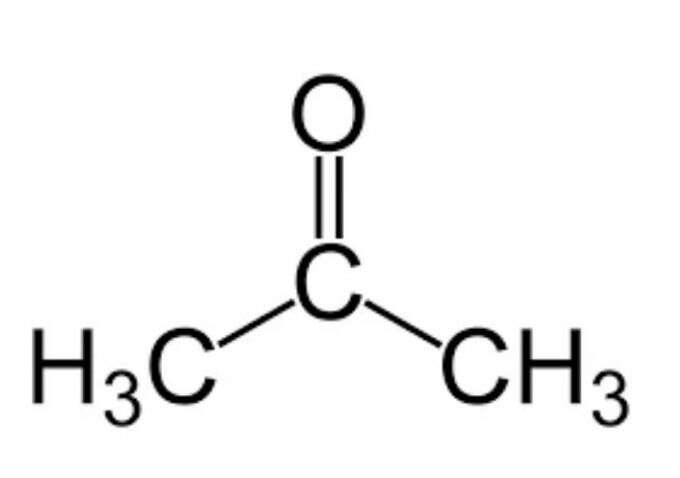
✓ Solved: Calculate the molarity and mole fraction of acetone in a 1.00-m solution of acetone (CH3 COCH3)...

SOLVED: The density of acetone is 791 kg/m³. This is equivalent to 3.60 x 10³ lb/L 1.74 x 10³ lb/L b) 3.60 x 10² lb/L 1.74 x 10⠴ lb/L A solid
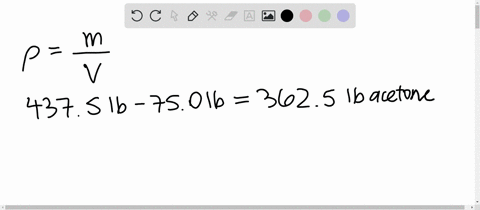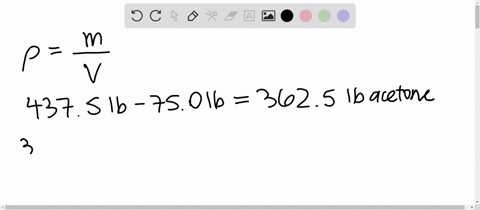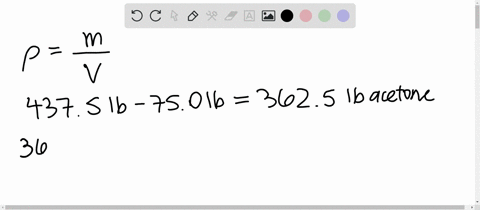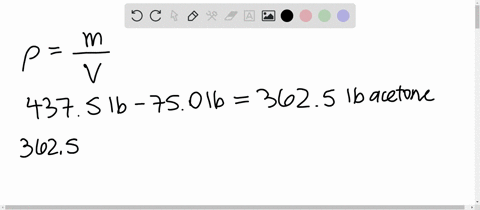
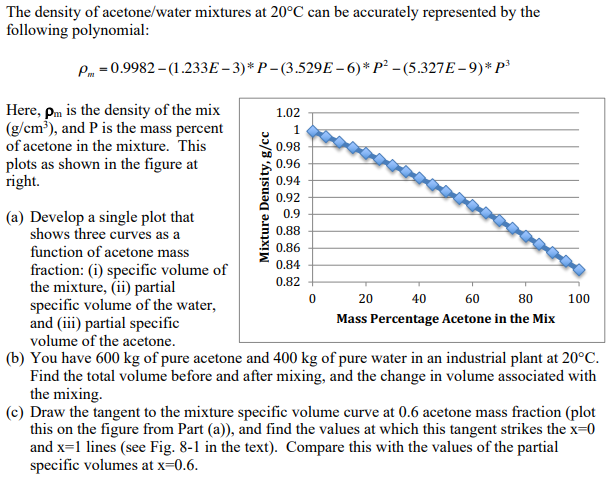
SOLVED: 1) Calculation of density of acetone in kg/m3 2) Estimate the density of a mixture of 50% acetone and 50% water by volume additivity

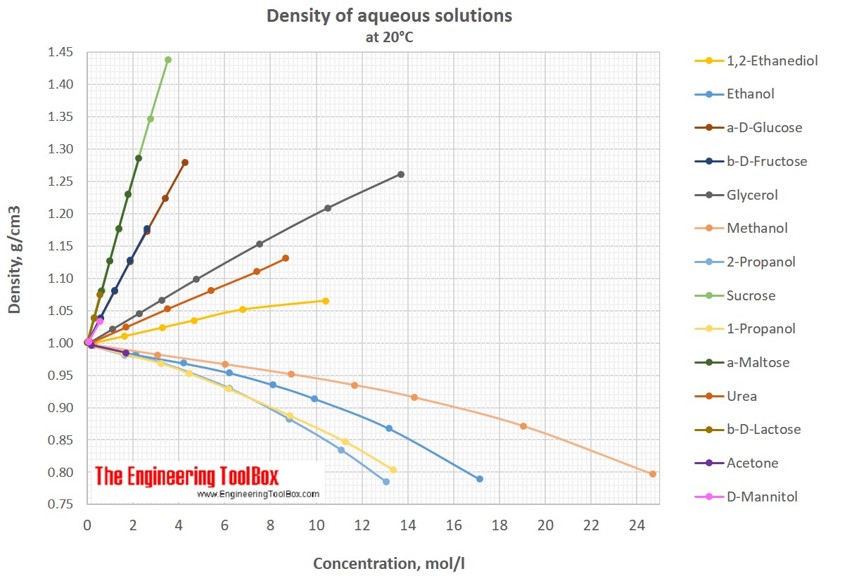
SOLVED: An aqueous solution of acetone (CH3COCH3) with a molality equal to 1.29 mol/kg has a density equal to 0.990 g/mL. What is the volume molar concentration?

OneClass: Acetone is a common solvent and has a density of 0.7899 g/mL. What volume of acetone, in mL...

Calculate the volumes of acetone and ethanol that was used to mix together in order to produce 1 liter (1000 mL) of the equimolar mixture. Ethanol Mr = 46 and a density (

High performance direct organic fuel cell using the acetone/isopropanol liquid organic hydrogen carrier system - ScienceDirect

SOLVED: 450 mL of acetone (density, Ï = 791 kg/m³) vapor is condensed at 56 °C. Ï hv for acetone is 30.2 kJ/mol. Mw for acetone is 58.08 g/mol. What is the